EnteroMedics Inc. (ETRM) got regulatory approval today for a first-of-its-kind weight-loss device that tricks users into feeling full. Now it has to persuade insurers to cover the gadget’s price tag of at least $15,000.
It’s a tough hurdle. Many insurers and government agencies have refused to cover previous weight-loss treatments, such as Vivus Inc. (VVUS)’s Qsymia and Belviq, by Arena Pharmaceuticals Inc. and Eisai Co. To get a better outcome, EnteroMedics will have to show the device, Maestro Rechargeable System, is safe enough.
Insurance coverage could help EnteroMedics tap a market of 20 million obese patients in the U.S., according to Chief Financial Officer Greg Lea. The Food and Drug Administration approved the system today for obese patients 18 and older who have at least one other weight-related condition, such as Type 2 diabetes -- the first obesity device approved in the U.S. since 2007.
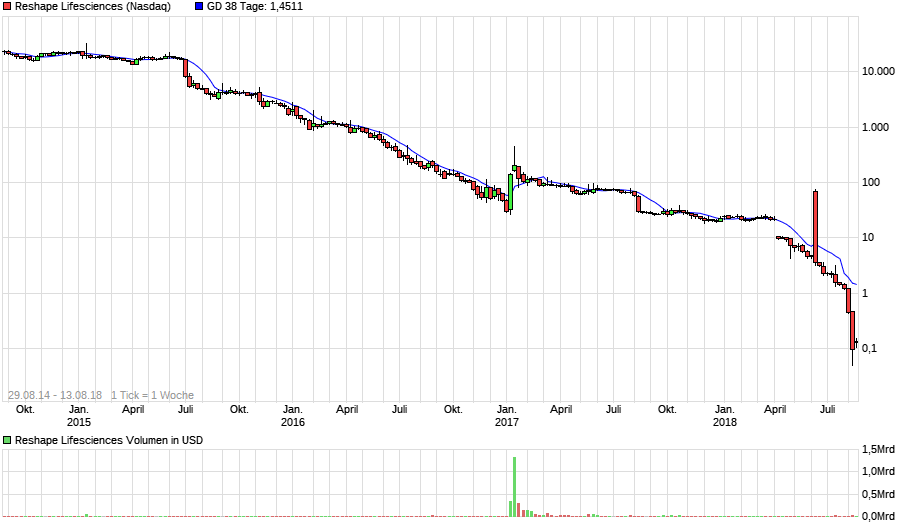
The FDA approval sent shares of EnteroMedics up 19 percent to $1.40 at the close, giving the company a market value of $97 million. The St. Paul, Minnesota-based company, founded in 2002, focuses on regulating the vagus nerve, which controls appetite.
Maestro is currently considered a Category 3 device by the FDA, with insufficient data to receive reimbursement by government agencies. EnteroMedics is pushing to have it classified as safe by next year, which would make private insurers more likely to come aboard, Lea said today in an interview.
Source: EnteroMedics Inc. via Bloomberg
EnteroMedics’ system uses electrodes implanted in the abdomen to send electrical pulses... Read More
“Our strength for the reimbursement path is, one, safety; we have a very strong benefit-versus-risk profile,” Lea said. “We don’t produce a lot of very severe issues with our procedure that create a million-dollar patient.”
Out of Pocket
In the meantime, EnteroMedics will try to introduce the treatment to patients who can pay out-of-pocket and don’t want to wait for insurance approval, Lea said. He estimated that the full price for Maestro, which targets the pathway between the brain and the stomach that controls feelings of hunger, could come in between $15,000 and $30,000. That’s competitive with comparable surgery options, according to Lea.
Maestro is likely to get insurance reimbursement because it fills an important gap, according to Chris Lewis, an analyst at Roth Capital Partners. He’s one of four who recommend buying the stock, while one analyst has a hold rating, according to data compiled by Bloomberg.
“There are a lot of patients in this gap between drugs and exercise and changes in diet, and on the other spectrum is the more invasive bariatric surgical options like Lap-Band,” Lewis said. “This fills that gap.”
Source: EnteroMedics Inc. via Bloomberg
EnteroMedics Inc.'s weight-loss device uses implantable electrodes to trick the brain... Read More
Getting government health agency approval by 2016 is still a “best-case scenario,” Lewis said.
“It’s not going to be a billion-dollar revenue company -- it’s going to take time to get there,” he said.
To contact the reporter on this story: Doni Bloomfield in New York at mbloomfiel12@bloomberg.net
To contact the editors responsible for this story: Crayton Harrison at tharrison5@bloomberg.net John Lear
|


 Thread abonnieren
Thread abonnieren